Lorlatinib
Impurities
- Lorlatinib is approved in the US and in Europe for the second- or third-line treatment of ALK-positive metastatic non-small-cell lung cancer (NSCLC). Reference standards of Lorlatinib API,and its pharmacopeial, non pharmacopeial impurities, and stable isotopes are listed below.
stdClass Object
(
[pname] => Lorlatinib
[catalogue_number] => PA 12 1490000
[category_ids] => ,78,75,70,76,82,
[chemical_name] =>
[weight] => 406.42
[form] => C21H19FN6O2
[cas] => 1454846-35-5
[pslug] => 1454846-35-5-lorlatinib-api-pa121490000
[latest_product] => 0
[linkproducts] => 0
[offers_id] =>
[offers_name] =>
[offers_status] =>
[offers_start_date] =>
[offers_end_date] =>
[pageview] =>
[offers_slug] =>
[offers_product_id] =>
[offers_product_code] =>
[offers_master_id] =>
[offer_percentage] =>
[offers_product_main_cat] =>
)
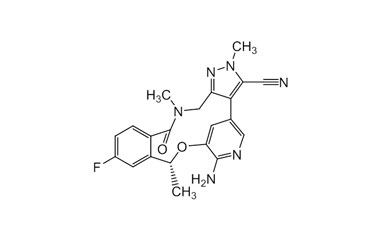
Catalogue No.:PA 12 1490000
Molecular Formula : C21H19FN6O2
Molecular Weight : 406.42
stdClass Object
(
[pname] => (S)-Lorlatinib
[catalogue_number] => PA 12 1491000
[category_ids] => ,78,75,70,76,82,
[chemical_name] =>
[weight] => 406.42
[form] => C21H19FN6O2
[cas] => 1454847-76-7
[pslug] => 1454847-76-7-s-lorlatinib-pa121491000
[latest_product] => 0
[linkproducts] => 0
[offers_id] =>
[offers_name] =>
[offers_status] =>
[offers_start_date] =>
[offers_end_date] =>
[pageview] =>
[offers_slug] =>
[offers_product_id] =>
[offers_product_code] =>
[offers_master_id] =>
[offer_percentage] =>
[offers_product_main_cat] =>
)
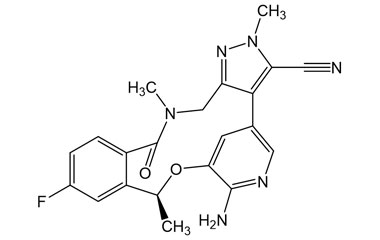
Catalogue No.:PA 12 1491000
Molecular Formula : C21H19FN6O2
Molecular Weight : 406.42