cabozantinib
Bulk Speciality Chemicals
- Cabozantinib is used in two forms. A capsule form is used since 2012 to treat medullary thyroid cancer and a tablet form is used since 2016 as a second line treatment for renal cell carcinoma.. Reference standards of Cabozantinib API, and its pharmacopeial, non pharmacopeial impurities, and stable isotopes are listed below
stdClass Object
(
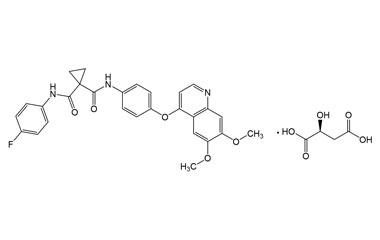
[pname] => Cabozantinib L-Malate Salt (BSC)
[catalogue_number] => PA BSC 06230258
[category_ids] => ,169,
[chemical_name] =>
[weight] => 635.6
[form] => C32H30FN3O10
[cas] => 1140909-48-3
[pslug] => 1140909-48-3-cabozantinib-l-malate-salt-bsc-pabsc06230258
[latest_product] => 0
[linkproducts] => 0
[offers_id] =>
[offers_name] =>
[offers_status] =>
[offers_start_date] =>
[offers_end_date] =>
[pageview] =>
[offers_slug] =>
[offers_product_id] =>
[offers_product_code] =>
[offers_master_id] =>
[offer_percentage] =>
[offers_product_main_cat] =>
)
Cabozantinib L-Malate Salt (BSC)