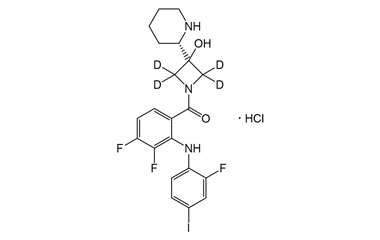
cobimetinib and its Impurities
It has been approved in Switzerland and the US, in combination with vemurafenib for the treatment of patients with unresectable or metastatic BRAF V600 mutation-positive melanoma.. Reference standards of Cobimetinib API, and its pharmacopeial, non pharmacopeial impurities, and stable isotopes are listed below